Earlier this year, David Hodl and I participated in the Phacilitate Cell and Gene Therapy Automation Special Interest Group (SIG) meeting where the group identified and addressed some of the biggest hurdles facing cell and advanced therapy companies on their path to industrializing complex medicines.
At present, cell therapy manufacture is a strange hybrid between highly sophisticated science and cottage industry. It takes several highly skilled individuals to make a therapy for a single person; whereas to meet the demand, and to be viable, one individual needs to be able to simultaneously produce “doses” for many people. As I see it, this is the primary purpose of mechanization and automation.
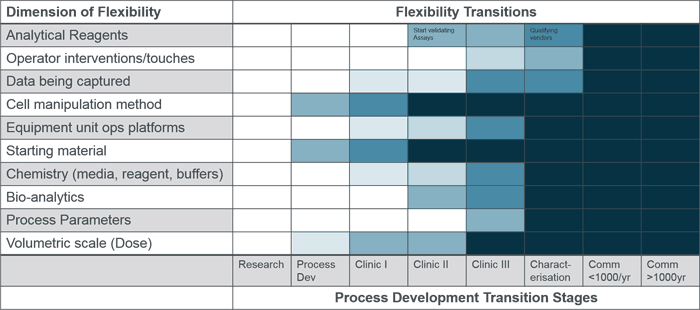
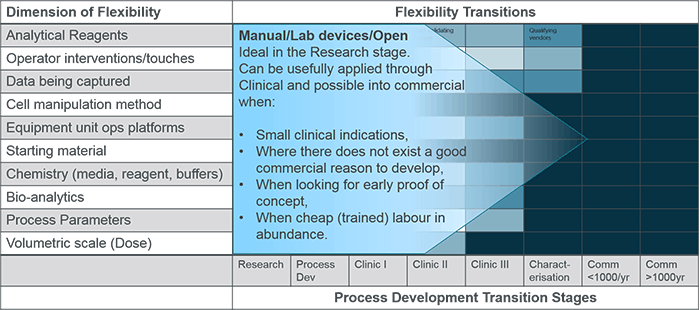
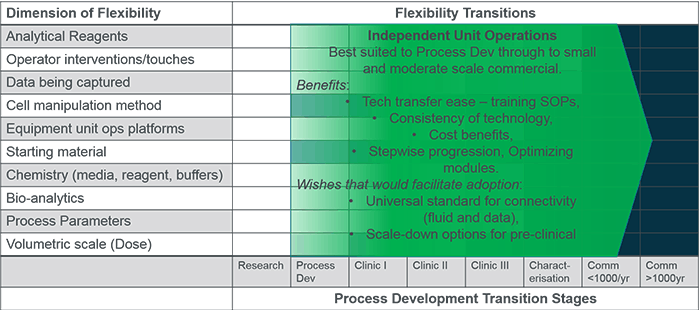
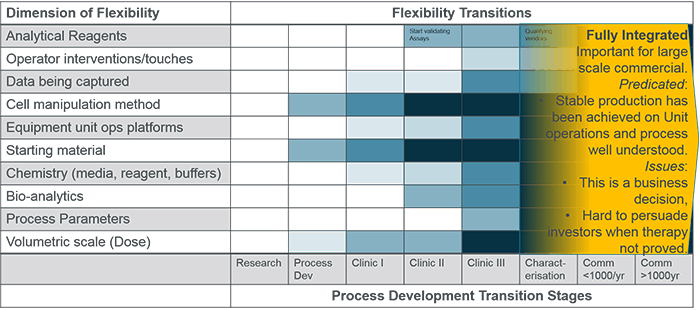
One such challenge faced by therapy product developers is how to choose production equipment that is both suitable for the current stage of development and will enable smooth transfer to commercial manufacture. During the Phacilitate Automation SIG, I facilitated a working session that addressed this question and the report is published below (with the permission of Phacilitate). The full report of all 12 working sessions can be downloaded here.